
Insights from recent episode analysis
Audience Interest
Podcast Focus
Publishing Consistency
Platform Reach
Insights are generated by CastFox AI using publicly available data, episode content, and proprietary models.
Est. Listeners
Based on iTunes & Spotify (publisher stats).
- Per-Episode Audience
Est. listeners per new episode within ~30 days
1,001 - 10,000 - Monthly Reach
Unique listeners across all episodes (30 days)
5,001 - 25,000 - Active Followers
Loyal subscribers who consistently listen
501 - 5,000
Market Insights
Platform Distribution
Reach across major podcast platforms, updated hourly
Total Followers
—
Total Plays
—
Total Reviews
—
* Data sourced directly from platform APIs and aggregated hourly across all major podcast directories.
On the show
Recent episodes
SE4:E8: From Discovery to Process, Part 2: Getting Ugi with it - scaling a 3CL protease inhibitor
Mar 23, 2026
Unknown duration
SE4:E7: From Discovery to Process, Part 1: Building a 3CL Protease Inhibitor Through Smart Fluorine Placement and Design Intuition
Feb 2, 2026
Unknown duration
SE4:E6 - Mechanism-Driven Innovation in Process Research: Yining Ji Chen’s Road as a Rising Star
Nov 10, 2025
Unknown duration
SE4:E5 - Evolution of a Green Chemistry Trailblazer
Oct 13, 2025
Unknown duration
SE4:E4 - MedChem Masters: Molecules, Mentors and Mindsets
Aug 19, 2025
Unknown duration
Social Links & Contact
Official channels & resources
Official Website
Login
RSS Feed
Login
| Date | Episode | Description | Length | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 3/23/26 |  SE4:E8: From Discovery to Process, Part 2: Getting Ugi with it - scaling a 3CL protease inhibitor SE4:E8: From Discovery to Process, Part 2: Getting Ugi with it - scaling a 3CL protease inhibitor | In Part 2 of our Pharm to Table deep dive on MK‑7845, Dani and LC shift from invention to implementation, welcoming process chemists Nastaran Salehi Marzijarani and Ben Turnbull to the pod. We explore how the team re‑imagined the synthesis to enable robust, scalable supply—and why a classic multicomponent reaction you probably remember from school, the Joullié–Ugi reaction, ended up stealing the show. Along the way, we unpack route selection trade‑offs, lifecycle thinking in process chemistry, and what it really takes to bring an antiviral to life at scale. Read and listen to some of the papers we discussed today:Asymmetric Synthesis of MK-7845, an Investigational Treatment for COVID-19 - Org. Lett.Part 1 of MK-7845 series: SE4:E7: From Discovery to Process, Part 1: Building a 3CL Protease Inhibitor Through Smart Fluorine Placement and Design IntuitionFollow the Pharm to Table podcast on X - @PharmtoTablePodVisit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 2/2/26 |  SE4:E7: From Discovery to Process, Part 1: Building a 3CL Protease Inhibitor Through Smart Fluorine Placement and Design Intuition SE4:E7: From Discovery to Process, Part 1: Building a 3CL Protease Inhibitor Through Smart Fluorine Placement and Design Intuition | In this first installment of our two‑part Pharm to Table special on MK‑7845, Dani and LC sit down with medicinal chemist Valerie Shurtleff to unpack the discovery journey behind this 3CL protease inhibitor—from early structural insights to an unexpectedly effective difluorobutyl isostere. Val walks us through how the team balanced potency, VLE, metabolic challenges, and design intuition to land on a well‑rounded preclinical candidate - one of the most dynamic programs of her career. Along the way, she reminds us why asking good questions, collaborating relentlessly, and occasionally surviving a “Hot Ones” challenge are all essential scientific skills. It’s an episode packed with chemistry, creativity, and just enough fluorine to keep things spicy.Stay tuned for Part 2, where we shift from invention to implementation and explore how the process chemistry team brought MK‑7845 to life at scale.Read some of the papers we discussed today:Invention of MK-7845, a SARS-CoV-2 3CL Protease Inhibitor Employing a Novel Difluorinated Glutamine Mimic - J. Med. Chem.Follow the Pharm to Table podcast on X - @PharmtoTablePodVisit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 11/10/25 |  SE4:E6 - Mechanism-Driven Innovation in Process Research: Yining Ji Chen’s Road as a Rising Star SE4:E6 - Mechanism-Driven Innovation in Process Research: Yining Ji Chen’s Road as a Rising Star | Join us for a conversation with Yining Ji Chen, recipient of the 2025 ACS Rising Star Award, on how mechanistic insight powers innovation in pharmaceutical process research. We dive into her career journey and standout projects—from Pd-catalyzed C–N coupling for HCV therapy to dynamic phosphorylation in nucleotide synthesis—plus her creation of LED‑NMR for real-time photochemical reaction monitoring. Hear how deep mechanistic understanding drives robustness and safety, why she moved to the Data‑Rich Measurements group, and which emerging analytical technologies excite her most. A celebration of scientific rigor shaping efficient, reliable pharma processes!Read some of the papers we discussed today:A multifunctional catalyst that stereoselectively assembles prodrugs - ScienceA rational pre-catalyst design for bis-phosphine mono-oxide palladium catalyzed reactions - Chem SciLED-Illuminated NMR Spectroscopy: A Practical Tool forMechanistic Studies of Photochemical Reactions - ChemPhotoChemFrom at-line to online NMR: coupling probe-based autosampler with benchtop NMR - Reaction Chemistry and EngineeringFollow the Pharm to Table podcast on X - @PharmtoTablePodVisit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 10/13/25 |  SE4:E5 - Evolution of a Green Chemistry Trailblazer SE4:E5 - Evolution of a Green Chemistry Trailblazer | In this episode, Dani and LC welcome Jeff Moore, recipient of the 2025 ACS Affordable Green Chemistry Award, to discuss his pioneering work in biocatalysis at Merck. The conversation explores the evolution (pun intended) of biocatalytic processes in pharmaceutical manufacturing, highlighting key milestones such as the adoption of transaminases and imine reductases, and their impact on drug synthesis and sustainability. Jeff shares stories from his career, including how being 'lazy' when doing science can lead to major innovations and the practical benefits of enzyme-driven chemistry. He even leaves us with insights into the future frontiers of biocatalysis, from cascade reactions to microfluidics.Read some of the papers we discussed today:Convergent Kilogram-Scale Synthesis of Dual Orexin Receptor Antagonist - Organic Letters Biocatalytic Asymmetric Synthesis of Chiral Amines from Ketones Applied to Sitagliptin Manufacture - ScienceEnabling Biocatalysis by High-Throughput Protein Engineering Using Droplet Microfluidics Coupled to Mass Spectrometry - ACS OmegaFollow the Pharm to Table podcast on X - @PharmtoTablePodVisit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 8/19/25 |  SE4:E4 - MedChem Masters: Molecules, Mentors and Mindsets SE4:E4 - MedChem Masters: Molecules, Mentors and Mindsets | In this episode, Dani and LC interview Dr. Shane Krska, recipient of the ACS Arthur C. Cope Award, and Dr. Petr Vachal, recipient of the ACS Earle B. Barnes Award for Leadership in Chemical Research Management. Shane shares his pioneering work in catalytic methods and high-throughput experimentation (HTE) for drug discovery, while Petr discusses his leadership role driving innovation at Merck. They reflect on their career journeys from graduate school to industry, emphasizing personal growth and the importance on taking on diverse roles. We wrap it up by asking Shane and Petr what has made them the most proud over their 20+ year careers - which spans from innovations in direct-to-biology for accelerating design-make-test to advancing peptides as a modality of the future!Read the papers we discussed today:The Catalysis Laboratory at Merck: 20 Years of CatalyzingInnovationThe medicinal chemist's toolbox for late stage functionalization of drug-like moleculesHighly Efficient Asymmetric Synthesis of SitagliptinFollow the Pharm to Table podcast on X - @PharmtoTablePodVisit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 5/19/25 |  S4:E3 - London Calling ... Spirocycles S4:E3 - London Calling ... Spirocycles | This month we sit down with Damien Valette, Olivia Boyd and David Petrone to chat about their latest paper in Organic Letters on the desymmetrization of prochiral dienones to control all-carbon quaternary centers. This reaction leverages biocatalysis, specifically an ene-reductase [ERED] enzymes (which are making their second appearance on the pod - see S1:E5).David, Damien and Olivia walk us through the discovery of the reaction and why it's uniquely suited to set these challenging stereocenters. This work was done in the context of work at the med-chem/process interface which we explore as well.Damien and Olivia hail from our London UK MSD site and give us details on the progress of our company's build in the region as well as chat with us about how to be successful when collaborating with scientists on another continent (David).Read the papers we discussed today:HTE-Enabled Development of an Ene-Reductase-Catalyzed Desymmetrization: Remote Control of All-Carbon Quaternary γ-Centers - Org Lett 2024Follow the Pharm to Table podcast on X - @PharmtoTablePodVisit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 4/21/25 |  S4:E2 - Making Sulfur Smell like Success! S4:E2 - Making Sulfur Smell like Success! | Join us on the Pharm to Table podcast for a delightful chat with Ethan Wappes from our South San Francisco site, as we dive into the exciting world of thiophene synthesis! Ethan is a member of our Discovery Process Chemistry group and worked alongside medicinal chemists to devise a better synthetic route towards substituted thiophenes. Ethan shares insights from his recent Org. Lett. on a novel Rh-catalyzed [3+2] method that opens the door to highly substituted dihydrothiophenes, enhancing our medicinal chemistry toolkit. We explore how this strategy not only simplifies complex thiophene syntheses but also paves the way for new drug development possibilities!Read the papers we discussed today:Fully Substituted Thiophene Synthesis via (3 + 2) with Thiadiazoles - Org Lett 2024Follow the Pharm to Table podcast on X - @PharmtoTablePodVisit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 11/18/24 |  S3:E10 - Look what ADCs made us do S3:E10 - Look what ADCs made us do | Antibody drug conjugates, or ADCs, are all the rage in the industry right now and chemists play an important role in their development. This month we sat down with Serge Ruccolo and Marion Emmert to talk about their papers in this area. Their work addresses two main challenges of ADC development: 1) potent handling of cytotoxic payloads and 2) selective unmasking of cysteine residues to enable conjugation to the antibody. Tune in to hear how one develops these chemistries; How electrochemistry and microfluidics can be useful tools for the chemistry of these important molecules. You'll learn a ton, we did! Read the papers we discussed today: “Build Your Own” ADC Mimics: Identification of Nontoxic Linker/Payload Mimics for HIC-Based DAR Determination, High-Throughput Optimization, and Continuous Flow Conjugation - OPRD Electrocatalytic Reduction of Disulfide Bonds across Chemical Modalities - Org Lett Follow the Pharm to Table podcast on X - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 10/17/24 |  S3:E9 - Surfing peptide production by going with the flow S3:E9 - Surfing peptide production by going with the flow | Ready to go with the flow? Then this episode is for you! In this episode, we dive into the exciting world of Continuous-Flow Solid-Phase Peptide Synthesis (CF-SPPS) with special guests Kyle Ruhl and Mike DiMaso. We kick off the episode with a brief overview on various techniques used to synthesize peptides and how each method fits within the drug discovery/development timeline. From there, we serve up their recent paper on leveraging flow to synthesize peptides - exploring the challenges they faced, such as accommodating resin swelling and optimizing synthesis times. We do our best to shield you from any discussions about math when they dive into linear velocity which helped drive peptide synthesis optimization. Discover how CF-SPPS is unleashing rapid, multigram peptide deliveries, culminating in a remarkable 20-gram yield of a 10-mer peptide in just 4 hours! Read the paper we discussed today: Continuous-Flow Solid-Phase Peptide Synthesis to Enable Rapid, Multigram Deliveries of Peptides Follow the Pharm to Table podcast on X - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 9/16/24 |  S3:E8 - So you think you want to start a chemistry podcast? S3:E8 - So you think you want to start a chemistry podcast? | Join Dani and LC as they peel back the layers on podcasting in chemistry. This insightful episode dives into their recently published Organic Letters editorial "Should You Start a Chemistry Podcast?" with special contributor and corresponding author Matt Horwitz from Synthesis Workshop sharing his unique perspective. But that's not all - get ready for an artistic twist as Brendan Burkett from Chemscrapes reveals how he transforms chemistry concepts into visually striking designs for the chemistry community (including CEN!). Whether you're an aspiring podcaster, a chemistry communicator, or just someone fascinated by the interplay of science and art, this episode promises an engaging exploration. Tune in for a candid discussion blending personal experiences, creative insights, and a passion for making chemistry more accessible to all. Don't miss this multimedia showcase!" Follow the Pharm to Table podcast on X - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table Org. Lett. featured: https://pubs.acs.org/doi/full/10.1021/acs.orglett.3c04181 | — | ||||||
Want analysis for the episodes below?Free for Pro Submit a request, we'll have your selected episodes analyzed within an hour. Free, at no cost to you, for Pro users. | |||||||||
| 8/5/24 |  S3:E7 - Your Rx for a Pharma Job: Insider Secrets (Part 2) S3:E7 - Your Rx for a Pharma Job: Insider Secrets (Part 2) | Time for the main course! Our #MerckChemistry hiring experts are dishing out more savory tips to help you get hired in pharma. In this second part, they'll guide you through mastering the job talk, nailing those crucial on-site interviews, and evaluating offers strategically. Learn how to deliver an engaging presentation, handle tough questions, and leave a lasting impression. Get advice on navigating those 1:1 interviews with poise. Once you've aced the interviews, find out how to weigh compensation packages and negotiate the most appetizing offer for your career growth. Whether you're a fresh grad looking to land that first gig or a seasoned pro ready for the next opportunity, this episode is the secret sauce you need! Follow the Pharm to Table podcast on X - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 6/5/24 |  S3:E5 - When Robots Work-It-Up! S3:E5 - When Robots Work-It-Up! | Alexandra Sun and Harrison Rose join the pod to share their work on a vision-guided automated liquid-liquid extraction (LLE) platform. That's right, before our robot overlords overtake us, they are going to help chemists and engineers with work-ups first! Even though it's often taken for granted, liquid-liquid extractions are part of virtually every chemical reaction for product isolation. Developing these unit operations is critical to chemists and engineers who design processes and Harrison and Alex's new tool can help expedite this. We also asked them for their secret to developing the best phase cuts! We learned a lot on this episode, and we hope you do too! Read the papers we discussed today: Vision-Guided Automation Platform for Liquid–Liquid Extraction and Workup Development - OPRD General Principles and Strategies for Salting-Out Informed by the Hofmeister Series - OPRD Follow the Pharm to Table podcast on X - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 4/22/24 | 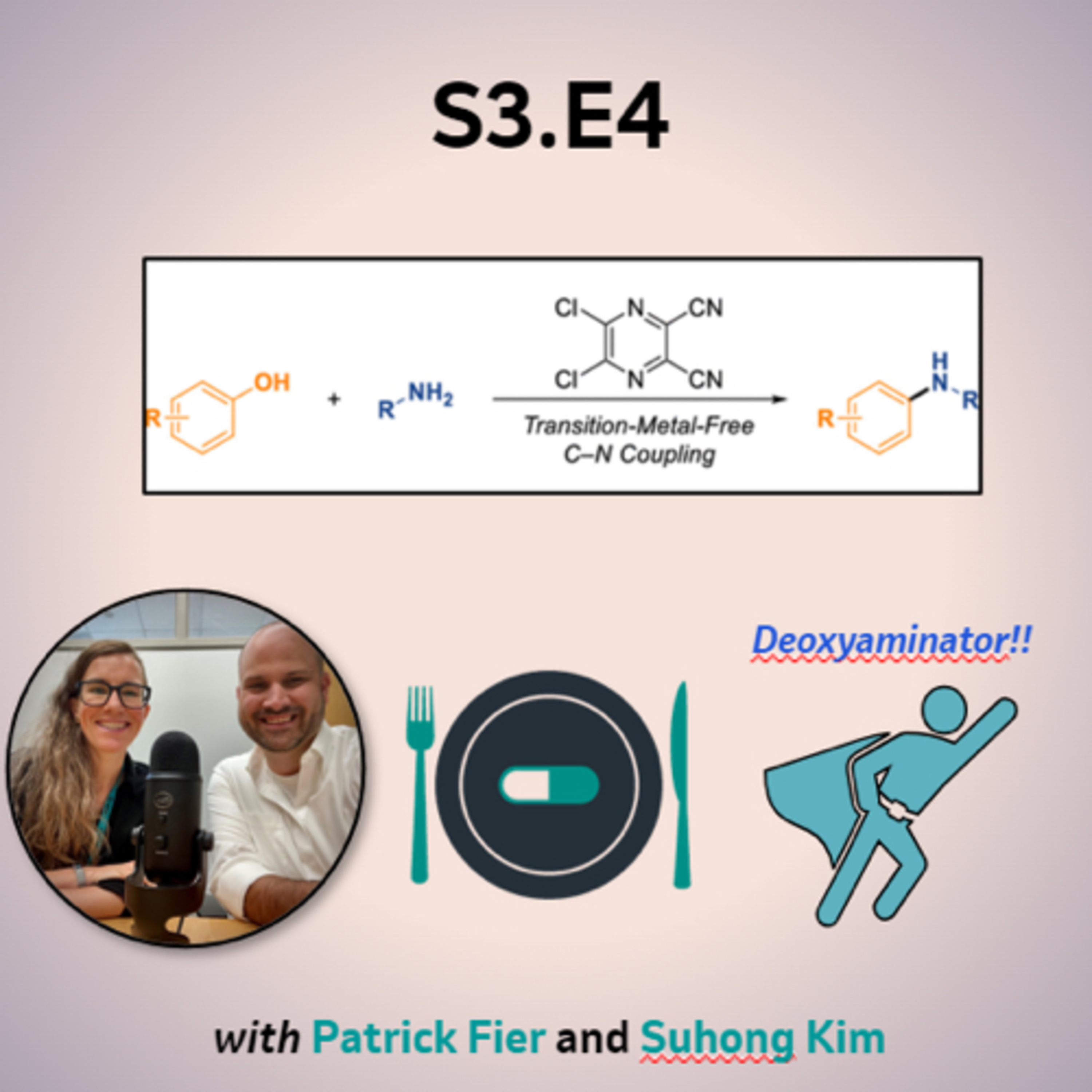 S3:E4 - "Asta La Vista, Baby" - The Aminator S3:E4 - "Asta La Vista, Baby" - The Aminator | Patrick Fier and Suhon Kim join the pod to share their lates collaboration on a new C-N cross-coupling protocol which leverages a novel multifunctional template reagent. The work emerged from a side-project that started many years ago on the creation of the "Aminator" reagent which inspired the latest reaction. The couplings have very broad scope and Suhong and Patrick explain their motivation for pursuing the work and insights about the reaction enabled by this reagent. We learned a lot on this episode, and we hope you do too! Read the papers we discussed today: Transition-Metal-Free C–N Cross-Coupling Enabled by a Multifunctional Reagent - JACS A Multifunctional Reagent Designed for the Site-Selective Amination of Pyridines - JACS Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 2/20/24 |  S3:E3 - A chemist, a physicist and an enzyme walk into a bar... S3:E3 - A chemist, a physicist and an enzyme walk into a bar... | Nadine Kuhl (process chemist) and Jacob Forstater (engineer and physicist) join the pod to share a pretty neat story that had its origins from a chemical catalog brochure advertising a new and green solvent called Cyrene. From that brochure blossomed a really amazing biocatalytic transamination to make a key chiral amine building block. In order to make this process more robust and tolerant of organic solvents, the team immobilized the transaminase enzyme and leveraged a Spinchem rotating bed reactor (we also had no idea what this was, but now we do) to make a robust process. We learned a lot on this episode, and we hope you do too! Read the papers we discussed today: Amination of a green solvent via immobilized biocatalysis for the synthesis of nemtabrutinib - ACS Catalysis Utilizing biocatalysis and a sulfolane-mediated reductive acetal opening to access nemtabrutinib from cyrene - Green Chemistry Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 1/15/24 |  S3:E2 - Barton-McCombie Beware! S3:E2 - Barton-McCombie Beware! | Charles Yeung (@Charles_chem) joins the pod to share some of the latest results from a collaboration with Prof. Wickens' lab (@WickensGroup) at the University of Wisconsin Madison. In their latest paper published in Angewandte Chemie, they disclose a practical and general protocol for alcohol dehydrogenation - putting the Barton-McCombie on notice! The work was supported by a Merck post-doctoral fellow, Myriam Mikhael (@MyriaMikhael), who shares her experience as an industrial PDF. Read the papers we discussed today: Practical and General Alcohol Deoxygenation Protocol - ACIE Translating Planar Heterocycles into Three-Dimensional Analogs by Photoinduced Hydrocarboxylation - ACIE Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 12/11/23 |  S3.E1: All I Want for the holidays are cross-couplings, collaborations and cookies!! S3.E1: All I Want for the holidays are cross-couplings, collaborations and cookies!! | For our special Holiday extravaganza, Dani and LC share their holiday plans before diving into the cookie jar of questions from some of our favorite listeners! How is R&D in industry similar to and different from how research is done in academia? (@DrSauce96) What is the most annoying academic misconception about work in the pharmaceutical industry? (@Teachforaliving) What things do you wish you knew before starting in industry? (@lifeof_dan) What is your favorite coupling reaction to run? Doesn’t have to be one you use most or the one with the broadest scope, just the one you look forward to setting up. (@ndchiappini) What can be done to help educate PhD students about industrial careers? The majority of them won’t go to academia, but it seems many profs know very little about non-academic pathways? (@Chem_Walker) So top off that nog and join us for a Holiday Snack! Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 7/17/23 |  S2.E8: Peptides, Cross-Electrophile Coupling and Maple Syrup! S2.E8: Peptides, Cross-Electrophile Coupling and Maple Syrup! | For this month's episode, Dani and LC serve up a recently published JACS article on Ni-catalyzed pyridinium cross-electrophile coupling for non-canonical amino acid synthesis and peptide late-stage functionalization which was done in collaboration with Professor Mary Watson’s group. In this episode, Dani and LC share the importance of investing in non-canonical amino acid chemistry with #MerckChemistry scientists Amélie Dion and Dipa Kalyani. We discuss what drew them to pyridinium cross-electrophile coupling and how Professor Watson’s expertise enabled the extension of this chemistry towards amino acid and peptide diversification via lysine deamination. By leveraging Dipa’s expertise in Discovery and high-throughput experimentation (HTE) – the team was able to create reaction generality for range of pharma relevant heteroaryl halides. In addition, Amélie was able to show that the lysine-derived pyridiniums survived solid-phase peptide synthesis and that the Ni-catalyzed cross-electrophile coupling could be achieved on-resin (rare!) – directly fitting into traditional medchem workflows. Professor Watson shares her insights on how working outside one’s area of expertise spurs innovation and ultimately what made this collaboration so successful. Many aspects of this collaboration were unique, which included real-time data sharing with our Discovery colleagues in addition to the team being largely composed of women! #WomenInChemistry Read the paper we discussed today here: Diversifying Amino Acids and Peptides via Deaminative Reductive Cross-Couplings Leveraging High-Throughput Experimentation - JACS Beyond 20 in the 21st Century: Prospects and Challenges of Non-canonical Amino Acids in Peptide Drug Discovery - ACS Med. Chem. Lett. Are Heterogeneous Catalysts Precursors to Homogeneous Catalysts? - JACS Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 6/15/23 |  S2.E7: So Far, So Cyanide: Green, air-tolerant and robust cyanation reactions. S2.E7: So Far, So Cyanide: Green, air-tolerant and robust cyanation reactions. | For this month's episode, Dani and LC go all in on Ni-catalyzed cyanations with Caleb Hethcox who recently published 2 papers on the topic - one with a phenomenal intern, Geraldo Duran-Camacho. Caleb shares how the internal team first identified this challenge and the solutions they explored to replace precious metal catalysts for cyanation reactions! Ultimately, the team identified a Ni-catalyzed system (go base metals!) and depending on the application, different reduction conditions which led to the different publications. As part of Merck's Future Talents Program, Caleb was joined by Geraldo who came to Merck and worked in our laboratories on this project. In addition, Geraldo gives us some of his learnings and insights from the experience which will be of great interest to potential future interns who listen to this episode. Read the paper we discussed today here: Nickel-Catalyzed Cyanation of (Hetero)aryl Bromides Using DABAL-Me3 as a Soluble Reductant - Org. Lett. Air-Tolerant Nickel-Catalyzed Cyanation of (Hetero)aryl Halides Enabled by a Green Reductant - J. Org. Chem. Merck Future Talent Program Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://podcasters.spotify.com/pod/show/pharm-to-table | — | ||||||
| 2/28/23 |  S2.E4: The Total Synthesis of Darobactin A (or is it Davobactin?) S2.E4: The Total Synthesis of Darobactin A (or is it Davobactin?) | For this month's episode, Dani and LC dive into the challenges of strained cyclic peptide synthesis with Niki Patel and Dave Petrone who recently completed the total synthesis of Darobactin A in collaboration with Professor Sarlah's group at UIUC. Now this wasn't Merck's first foray into natural product total synthesis, in fact Darobactin A is part of a rich legacy of total syntheses at Merck that spans 80 years! Niki and Dave share how the internal Merck team first envisioned putting this molecule together - including how to construct the complex non-canonical amino acids and ultimately how to forge the highly strained bis-macrolactams. They then walk us through how the team ended up collaborating with Professor Sarlah's team at UIUC combining efforts to complete the total synthesis. Niki, Dave, and Professor Sarlah describe that first meeting and share how they ultimately overcame the macrocyclization challenges which resulted in the total synthesis of Darobactin A! This episode is filled lots of learnings which include how to construct strained cyclic peptide but more importantly, how a strong network can elevate problem solving and potentially lead to a fruitful academic-industrial collaboration. Read the paper we discussed today here: Total Synthesis of Darobactin A - JACS Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://anchor.fm/pharm-to-table | — | ||||||
| 12/22/22 |  S2.E3: All I Want for Christmas Is Candy, Chainsaws, Chemistry and ANSWERS! S2.E3: All I Want for Christmas Is Candy, Chainsaws, Chemistry and ANSWERS! | For our special Holiday extravaganza, Dani and LC reminisce about the holidays while in grad school (think candy throw downs and chainsaws) before diving into the cookie jar of questions from some of our favorite listeners! What is a good way to figure out if you'd be a better fit for a medicinal chemistry or process chemistry role? (@billthechemist) What do you know now that you wished you knew during your undergraduate or early graduate school? (@MichaelAxiotes) How is pregnancy and parenthood handled in industry? (@EleanorMLandwe1) How do you decide with whom you collaborate with in academia? (@AnneJMcNeil) What recent chemistry breakthrough over the last few year has been the biggest surprise? (@AlexSolivan) If you could draw any chemical transformation on paper and have it work in the lab, what would it be and why? (@stanna_dorn) So top off that nog and join us for a Holiday Snack! Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://anchor.fm/pharm-to-table | — | ||||||
| 12/7/22 |  S2.E2: Enabling medicinal chemistry knowledge exchange with #ChemistryWizards (not trolls) S2.E2: Enabling medicinal chemistry knowledge exchange with #ChemistryWizards (not trolls) | For S2E2, we’re joined by drug hunting legends Doug Beshore and Izzat Raheem who recently co-authored a recent (and popular) paper in J. Med. Chem. on building a culture of knowledge exchange. Ironically, it’s easier to learn through the literature than from your organization, colleagues or group members - especially if colleagues have taken on different roles. As a result, building and maintaining a culture where prior knowledge is easily accessible and updatable is paramount to a healthy learning culture. In this episode we go into the 4 pillars that made knowledge exchange with Merck Discovery successful, which includes (1) IT; (2) knowledge capture; (3) participation and most importantly (4) culture. Knowledge management is not unique to industry - in fact, many of the lessons learned can be directly applied to an academic culture as well - and span beyond science and into DEI as well. So tune in to hear how simple practices can boost learning and knowledge exchange in your research. Read the papers we discussed today here: Building a Culture of Medicinal Chemistry Knowledge Sharing - J. Med. Chem. Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://anchor.fm/pharm-to-table | — | ||||||
| 11/1/22 |  S2.E1: No Bald Eagles We're Harmed in the Recording of this Podcast S2.E1: No Bald Eagles We're Harmed in the Recording of this Podcast | We kick off season 2 with a bang, talking about complex molecule synthesis with John McIntosh and Nastaran Salehi. Cyclic dinucleotides (CDNs) are a total synthesis fan's dream with 10 stereocenters (including 2 at phosphorus) arranged around a 13 membered macrocycle formed by 2 non-canonical nucleosides. These endogenous secondary messenger molecules have attracted attention because of their activation of the immune system via the STING pathway. We go into all aspects of the innovative biocatalytic cascade that the team designed for their synthesis, including: 1) How a bald eagle cyclic guanosine-adenosine synthase (cGAS) was engineered to form the macrocycle; 2) How cGAS was leveraged by medicinal chemists to greatly accelerate drug discovery; 3) How the team strung together and optimized a 4-enzyme cascade to deliver a CDN directly from nucleotide building blocks. Read the papers we discussed today here: A kinase-cGAS cascade to synthesize a therapeutic STING activator - Nature New Mechanism for Cinchona Alkaloid-Catalysis Allows for an Efficient Thiophosphorylation Reaction - J. Am. Chem. Soc. Follow Nastaran Salehi - @Nastaran198 Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://anchor.fm/pharm-to-table | — | ||||||
| 10/24/22 |  S2 Kickoff: Answering Your Questions & S2 Sneak Peak S2 Kickoff: Answering Your Questions & S2 Sneak Peak | For our kickoff of Season 2, LC and Dani get together (in person!) to reflect on what’s changed since the Pharm to Table pod started in late 2020, answer a listener question (only 9 months late!) on whether a postdoc is needed for an industrial job, and give a sneak peak on what’s to come for Season 2. Whether you listen in the car or in the lab - buckle up and don that PPE - as Season 2 is destined to be full of energy, laughs and awesome #MerckChemistry. Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://anchor.fm/pharm-to-table | — | ||||||
| 7/1/22 |  S1.E10: Hot Tub Flow Time Machine S1.E10: Hot Tub Flow Time Machine | For the 10th, and final course of season 1, Dani and LC jump in the hot tub flow time machine with Cecilia Bottecchia and Francois Levesque, co-recipient of the Peter Dunn Green Chemistry Award, and learn how to discover, develop and scale-up a photobromination in flow! But don't let this deceptively simple bromination fool you - this reaction turned out to be a full course meal of challenges! We go into all aspects of the reaction, including: 1) Why they decided to use light vs AIBN to facilitate this reaction; 2) How to design a photoreactor you can use on kilo scale; 3) How to design a robust reaction that can run on >50kg scale; 4) What does hot tub chemistry have to do with a radical bromination reaction. Read the paper we discussed today here: A Continuous Flow Visible-Light-Induced Benzylic Bromination - Organic Process Research and Development Elucidation of Reaction Mechanisms Using In Situ LED-NMR - Journal of Organic Chemistry Design of a Kilogram Scale, Plug Flow Photoreactor Enabled by High Power LEDs - Organic Process Research and Development Follow Cecilia Bottecchia - @cecibottecchia Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://anchor.fm/pharm-to-table | — | ||||||
| 5/9/22 |  S1.E9: ChemSpeed, Daft Punk and Twinkly Lights S1.E9: ChemSpeed, Daft Punk and Twinkly Lights | For the 9th course, Dani and LC do the robot dance with Melodie Christensen (@chemchristensen on Twitter) #MerckChemistry Enabling Technologies group. Melodie shares her unique career path with us, including her current PhD with @procastiprof - more >10 years after starting her career in pharma. Resident expert in reaction automation, robotics and self-optimizing reactions, Melodie shares her journey in high-throughput and data-rich experimentation. We discuss her award winning paper in JOC and how she was able to use some of the insights of this work to develop an autonomously optimized Suzuki reaction! Finally, Melodie schools us on her best smoothie recipes and three letter acronyms. Read the paper we discussed today here: Enantioselective Synthesis of α-Methyl-β-cyclopropyldihydrocinnamates - Journal of Organic Chemistry Data-science driven autonomous process optimization - Communications Chemistry The Evolution of High-Throughput Experimentation in Pharmaceutical Development and Perspectives on the Future - OPRD Automation isn't automatic - Chemical Science Follow Melodie Christensen - @chemchristensen Follow the Pharm to Table podcast on Twitter - @PharmtoTablePod Visit our website at https://anchor.fm/pharm-to-table | — | ||||||
Showing 25 of 43
Sponsor Intelligence
Sign in to see which brands sponsor this podcast, their ad offers, and promo codes.
Chart Positions
4 placements across 4 markets.
Chart Positions
4 placements across 4 markets.
